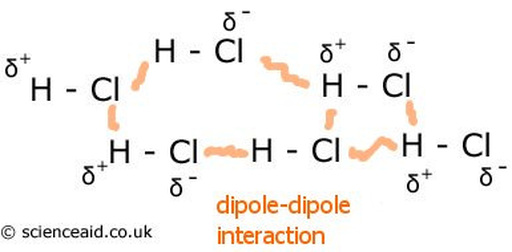
Diapole Diapole Forces
Diapole Diapole Forces are the attractions between oppositely charges regions of polar molecules.
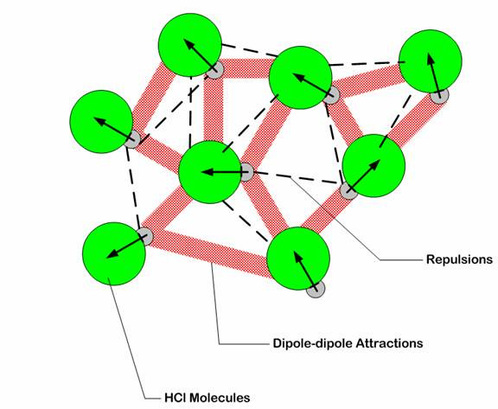
- they are stronger intermolecular forces than dispersion forces
- occur between polar molecules (that have permanent net dipoles)
- Although if the permanent net dipole within the polar molecules results from a covalent bond between a hydrogen atom and either fluorine, oxygen or nitrogen, the resulting intermolecular force is referred to
as a hydrogen bond
- occur between polar molecules (that have permanent net dipoles)
- Although if the permanent net dipole within the polar molecules results from a covalent bond between a hydrogen atom and either fluorine, oxygen or nitrogen, the resulting intermolecular force is referred to
as a hydrogen bond