Hydrogen Bonds
This is a special intermolecular force where the attraction occurs between a hydrogen atom and a higly electronegative atom with atleast one electron pair.
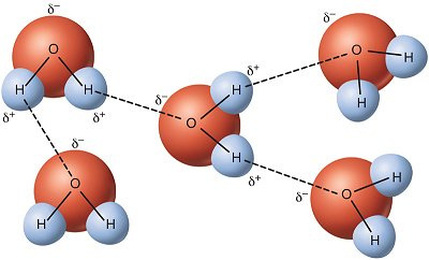
- hydrogen bonds occur between molecules that have a permanent net dipole.
- this means that hydrogen can bond with fluorine, oxygen or nitrogen. (as an example: water H2O or ammonia NH3)
- It is the strongest intermolecular force between all three types since the nucleus of hydrogen is very small and positively charged
while fluorine, oxygen and nitrogen are very electronegative resulting in the hydrogen atom being strongly attracted to them
- 'a highly localised positive charge on the hydrogen atom and highly negative localised charge on the fluorine, oxygen or nitrogen atom'. This means that the electrostatic attraction between these molecules is greater than polar molecules that dont have hydrogen covalently bonded to either fluorine, nitrogen or oxygen
- this means that hydrogen can bond with fluorine, oxygen or nitrogen. (as an example: water H2O or ammonia NH3)
- It is the strongest intermolecular force between all three types since the nucleus of hydrogen is very small and positively charged
while fluorine, oxygen and nitrogen are very electronegative resulting in the hydrogen atom being strongly attracted to them
- 'a highly localised positive charge on the hydrogen atom and highly negative localised charge on the fluorine, oxygen or nitrogen atom'. This means that the electrostatic attraction between these molecules is greater than polar molecules that dont have hydrogen covalently bonded to either fluorine, nitrogen or oxygen
