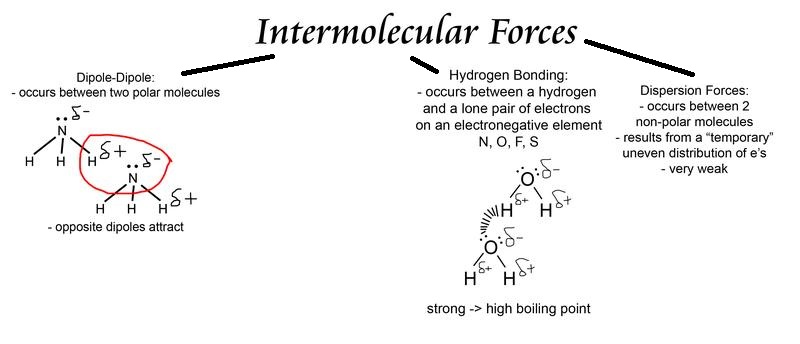
What are intermolecular forces?
When atoms are joined to one another they form a molecule that in turns has forces that bind them together. These forces are known as intermolecular forces.
- these forces play an important role in chemistry
- these forces are what determines the material's state of matter ( whether it is liquid, gas or solid) at a given tempreture
- they cause the solubalities of one substance in another
- although breaking an intermolecular attraction ( unlike breaking a chemical bond) does not change the chemical formula or chemical reactivity
- intermolecular forces determine if a reaction can occur and how quick is it
- these forces are what determines the material's state of matter ( whether it is liquid, gas or solid) at a given tempreture
- they cause the solubalities of one substance in another
- although breaking an intermolecular attraction ( unlike breaking a chemical bond) does not change the chemical formula or chemical reactivity
- intermolecular forces determine if a reaction can occur and how quick is it