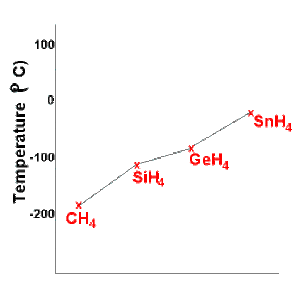
-RULE: Since melting or boiling result from a progressive weakening of the intermolecular attractive forces between the covalent molecules, the stronger the intermolecular force is, the more energy is required to melt the solid or boil the liquid.
-If only dispersion forces are present, then the more electrons the molecule has (and the more mass it has) the stronger the dispersion forces
will be, so the higher the melting and boiling points will be. As the mass of the molecules increases, so does the strength of the dispersion force acting between the molecules, so more energy is required to weaken the attraction between the molecules resulting in higher boiling points.
On the Other Hand
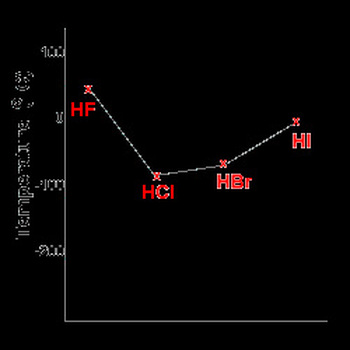
-As an example: the hydrides from group VII such as HF, HCL, HBr amd HI are polar
- If a covalent molecule has a permanent net dipole then the force of attraction between these molecules will be stronger than if only dispersion forces were present between the molecules. As a consequence, this substance will have a higher melting or boiling point than similar molecules that are non-polar in nature.
- In the hygrogen bond the hydrogen atom having a partial positive charge (H) and the halogen atom having a partial negative
charge (F, Cl, Br, I).
- Therefore the stronger the dipole-interactions acting between the molecules the higher the boiling point
Rule: as the molecular mass increases, the boiling point of
the hydrides increase.
- Although HF is an exception because of the stronger force of attraction between HF molecules resulting from hydrogen bonds acting between the HF molecules. Weaker dipole-dipole interactions act between the molecules of HCl, HBr and HI. So HF has a higher boiling point than the other molecules
in this series.
- In the hygrogen bond the hydrogen atom having a partial positive charge (H) and the halogen atom having a partial negative
charge (F, Cl, Br, I).
- Therefore the stronger the dipole-interactions acting between the molecules the higher the boiling point
Rule: as the molecular mass increases, the boiling point of
the hydrides increase.
- Although HF is an exception because of the stronger force of attraction between HF molecules resulting from hydrogen bonds acting between the HF molecules. Weaker dipole-dipole interactions act between the molecules of HCl, HBr and HI. So HF has a higher boiling point than the other molecules
in this series.
Furthermore
In general like dissolves like:
non-polar solutes dissolve in non-polar solvents
polar solutes will dissolve in polar solvents such as water and glucose
non-polar solutes dissolve in non-polar solvents
polar solutes will dissolve in polar solvents such as water and glucose