Dispersion Forces
Dispersion forces or London forces are weak forces that result from temporary shifts in density of the electrons that are found in the electron cloud and due to the temparay nature of the diapoles.
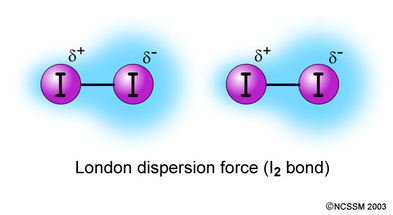
- dispersion forces exist between non polar molecules and noble gas atoms ( for example argon and octane)
- These types of attractive forces are caused by a phenomenon known as
instantaneous dipole formation. In this process, electron distribution in
the individual molecules suddenly becomes asymmetrical, and the newly formed
dipoles become attracted to one another
- When two non-polar molecules are in contact:
* the electron density around the nucleus ( in each molecule) is greaterin one region of each cloud
* each molecule forms a temportary dipole
- a weak dispersion force is present between oppositely charges regions of the diapoles when the temporary diapoes are close to one another
-
- These types of attractive forces are caused by a phenomenon known as
instantaneous dipole formation. In this process, electron distribution in
the individual molecules suddenly becomes asymmetrical, and the newly formed
dipoles become attracted to one another
- When two non-polar molecules are in contact:
* the electron density around the nucleus ( in each molecule) is greaterin one region of each cloud
* each molecule forms a temportary dipole
- a weak dispersion force is present between oppositely charges regions of the diapoles when the temporary diapoes are close to one another
-
